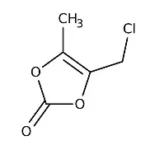
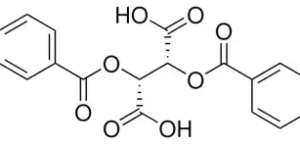
“We invest in our people and communities, fostering a culture of responsibility and care.”
G. Siva Rami Reddy
Director
G.DIneshwara Reddy
Partner
R&D Facilities
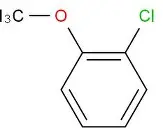
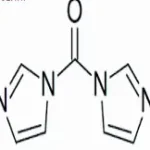
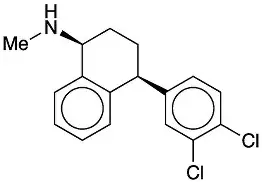
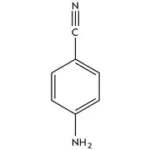
Suviro Life Sciences has set up a Research and Development Centre. It has helped us to make a successful entry into an enhanced product portfolio. The R&D Centre facility focuses on generic API and intermediates development. Our AR&D facility is located in Hyderabad.
QC and QA Facilities
As an organisation engaged in a business affecting human lives and upholding moral & ethical values, Suviro Life Sciences strongly believes in manufacturing of high quality products. Suviro aims to maintain and enhance the quality of the products being manufactured across the organisation through continuous process development as per regulatory and customer requirements.
Manufacturing Facilities
Suviro Life Sciences manufacturing facilities including Production blocks, Clean rooms, Warehouse and all utilities are established according to the GMP and FDA requirements and guidelines. We have all the required manufacturing drug licenses and certifications from the relevant authorities.
Request a Quote
Ready to Work Together? Build a project with us!